How activated alumina works
2023-05-18
Activated alumina is an important industrial catalyst widely used in many chemical reactions. Its working principle involves many disciplines such as physical chemistry, surface science and catalytic reaction. In this article, I will explain in detail how activated alumina works.
First of all, let's understand what is activated alumina. Activated alumina is a catalyst mainly composed of γ-Al2O3, which has the characteristics of high specific surface area, excellent thermal stability and chemical stability. It is usually made by drying aluminum hydroxide or other aluminum salts at high temperature.
The catalytic properties of activated alumina are closely related to its surface structure. In surface science research, people divide surfaces into lattice planes and non-lattice planes. The lattice plane refers to the surface directly in contact with the interior of the crystal, where the atomic arrangement is relatively regular and the surface energy is high. The non-lattice plane is formed by defects, boundaries and holes on the lattice plane, and its atomic arrangement is relatively loose and the surface energy is low. For catalytic reactions, the amorphous plane is the surface that plays a major role. Because catalytic reactions require substances to undergo adsorption, reaction, and dissociation processes on the surface, defects, boundaries, and holes on the non-lattice surface provide more active sites and orientations for these processes.
For activated alumina, its amorphous surface is mainly composed of holes and defects. The pores can increase the specific surface area of the catalyst and increase the contact rate between the reactant molecules and the catalyst. Defects can affect the electronic structure and acid-base properties of the catalyst, so that the catalyst has better catalytic activity and selectivity. In addition, there are some chemical groups, adsorption species and metal ions on the surface of activated alumina, which also play an important role in the catalytic reaction.
In the catalytic reaction, activated alumina mainly plays the following roles:
* Adsorption of reactants
Catalytic reactions first require reactants to be adsorbed on the surface of the catalyst to form adsorbed states or intermediates. Through adsorption, reactant molecules can come together to form a state where chemical reactions are more likely to occur.
* Activation of reactants
The reactant molecules adsorbed on the catalyst surface need to be activated so that they enter a more reactive state. This usually involves the breaking and recombination of certain bonds in the reactant molecules.
* Catalytic reactions
Catalysts facilitate chemical reactions once adsorbed states or intermediates are formed. It can act on the reactant molecules by providing acid, alkali or metal ions to change the electronic structure and chemical bonds of the reactant molecules and promote the reaction.
* Dissociation products
After the reaction is complete, the products need to dissociate from the catalyst surface. This process can also be affected by catalysts.
In summary, the working principle of activated alumina is to promote the occurrence of chemical reactions through the catalytic properties of its surface. The pores, defects, chemical groups, adsorbed species and metal ions on the surface of activated alumina provide abundant active sites and catalytic centers for catalytic reactions.
In different catalytic reactions, the specific mode of action of activated alumina is also different. For example, in the ethylene hydrogenation reaction, activated alumina mainly plays the role of mass transfer between the catalyst and the reactant, and realizes effective contact between the reactant and the catalyst through adsorption and dissociation of reactant molecules; in the styrene oxidation reaction, Activated alumina mainly plays the role of its acidity and transition metal ions to promote the activation and reaction of reactants.
In addition, activated alumina can also participate in a variety of reaction mechanisms, such as redox, acid-base neutralization, rearrangement and addition reactions. Its catalytic properties are affected by many factors, such as the physical and chemical properties of the catalyst, reaction conditions (such as temperature, pressure, reactant concentration, etc.), the morphology, surface structure and particle size of the catalyst, etc.
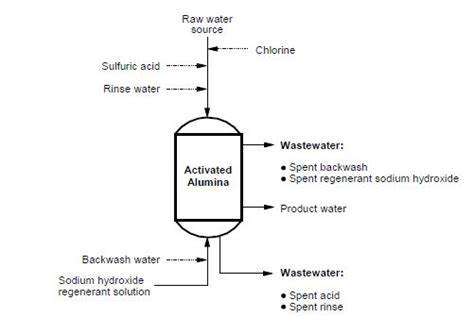
In conclusion, activated alumina is an important catalyst with high specific surface area, catalytic activity and selectivity. Its working principle involves multiple disciplines such as physical chemistry, surface science, and catalytic reactions. It promotes the occurrence of chemical reactions through active sites such as pores, defects, chemical groups, adsorbed species, and metal ions on the surface.

